Which of the following atoms can attract a hydrogen atom in a hydrogen bond.
If you’re looking for which of the following atoms can attract a hydrogen atom in a hydrogen bond images information connected with to the which of the following atoms can attract a hydrogen atom in a hydrogen bond keyword, you have pay a visit to the right blog. Our site always gives you hints for downloading the maximum quality video and picture content, please kindly search and locate more enlightening video content and images that match your interests.

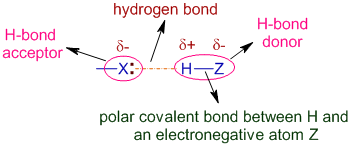
Such an interacting system is generally denoted DnHAc where the solid line denotes a polar covalent bond and the dotted or dashed line. The bond between Na and Cl in salt e. Hydrogen bond acceptor and the hydrogen itself can be as short as 18-19 Å well below the sum of the atomic radii eg. A hydrogen atom bonded to F O or N is attracted to an electron pair.
A hydrogen atom bonded to F O or N is attracted to an electron pair.
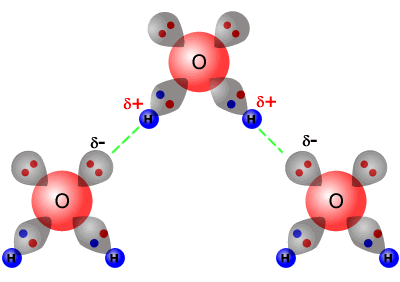
The bond between Na and Cl in salt e. Also the oxygen which has two lone pairs of electrons can form two hydrogen bonds with hydrogen atoms. In the picture of two water molecules at lower right the oxygen of the water molecule B is the hydrogen bond donor. Hydrogen bond strengths range from 4 kJ to50 kJ per mole of hydrogen bonds. The only way these atoms can get closer to one another is if.
 Source: slidetodoc.com
Source: slidetodoc.com
The bond between Mg and Cl in MgCl2 c. So hydrogen bonding is possible only in those compounds in which the hydrogen atom is directly bonded to fluorine oxygen or nitrogen. There are three basic ways that the outer electrons of atoms can form bonds. A hydrogen atom bonded to F O or N is attracted to an electron pair. The bond between Na and Cl in salt e.
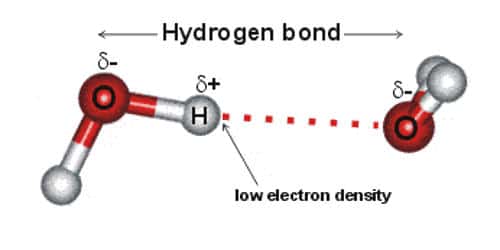
The hydrogen atoms are bound to the highly electronegative oxygen atom which also possesses two lone pair sets of electrons making for a very polar bond.
The nitrogen atom is called the hydrogen bond acceptor because it is accepting the hydrogen from the oxygen. Also the oxygen which has two lone pairs of electrons can form two hydrogen bonds with hydrogen atoms. Hydrogen bond is formed only by the three highly electronegative elements- fluorine oxygen and nitrogen. Two oxygen atoms c.
 Source: ibchem.com
Source: ibchem.com
Answer 1 of 13. Hydrogen bond is formed only by the three highly electronegative elements- fluorine oxygen and nitrogen. It results fromthe attractive force between a hydrogen atom covalently bonded toa very electronegative atom such as a N O or F atom and another veryelectronegative atom. These can then attract one another and create substances with higher boiling points.
 Source: pinterest.com
Source: pinterest.com
The partially positive hydrogen atom of one molecule is then attracted to the oxygen atom of a nearby water molecule see Figure below. The first way gives rise to what is called an ionic bond. Atom - atom - Atomic bonds. The bond between C.
 Source: in.pinterest.com
Source: in.pinterest.com
These can then attract one another and create substances with higher boiling points. A hydrogen atom in a molecule forms a bond with any atom. The only way these atoms can get closer to one another is if. You know that the Lennard-Jones potential rises with 1r12 if we get closer than the Van der Vaals radii.
Also the oxygen which has two lone pairs of electrons can form two hydrogen bonds with hydrogen atoms. The partially positive hydrogen atom of one molecule is then attracted to the oxygen atom of a nearby water molecule see Figure below. The bond between the H of one water molecule and the O of another water molecule b. The bond between Na and Cl in salt e.
Hydrogen bond is formed only by the three highly electronegative elements- fluorine oxygen and nitrogen.
As these three atoms are the only ones with sufficient electronegativity that when bound to hydrogen can form hydrogen bonds. Water shown above is a molecule which can exhibit hydrogen bonding. Atom - atom - Atomic bonds. So hydrogen bonding is possible only in those compounds in which the hydrogen atom is directly bonded to fluorine oxygen or nitrogen. Answer 1 of 13.
 Source: adichemistry.com
Source: adichemistry.com
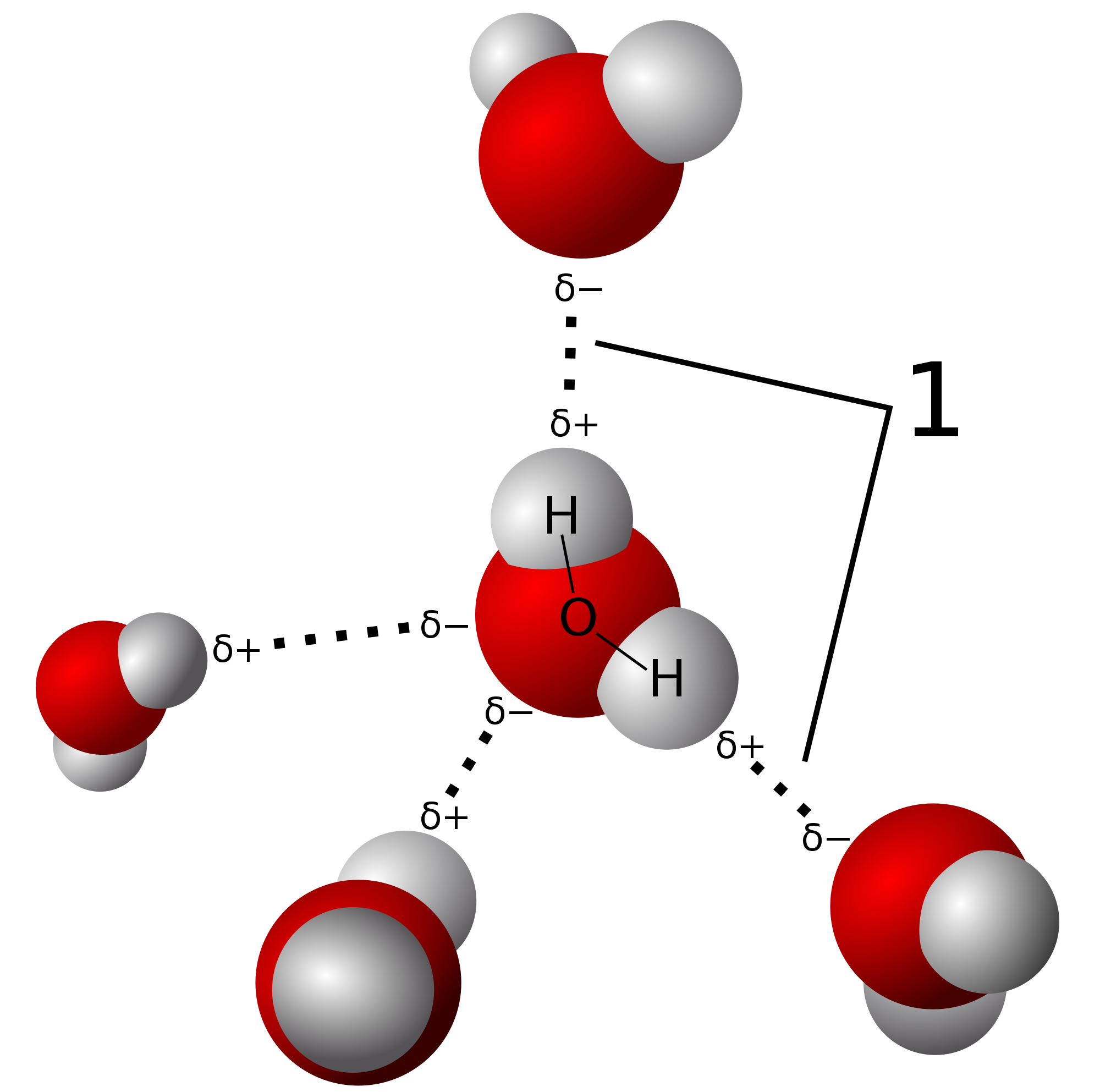
In the picture of two water molecules at lower right the oxygen of the water molecule B is the hydrogen bond donor. 12Å for hydrogen and 15Å for oxygen and nitrogen. A hydrogen atom forms a covalent bond with another atom. The only way these atoms can get closer to one another is if. Hydrogen bond is formed only by the three highly electronegative elements- fluorine oxygen and nitrogen.
In the picture of two water molecules at lower right the oxygen of the water molecule B is the hydrogen bond donor. The first way gives rise to what is called an ionic bond. A hydrogen bond can form between_____ adjacent to each other. It results fromthe attractive force between a hydrogen atom covalently bonded toa very electronegative atom such as a N O or F atom and another veryelectronegative atom.
12Å for hydrogen and 15Å for oxygen and nitrogen.
Once the way atoms are put together is understood the question of how they interact with each other can be addressedin particular how they form bonds to create molecules and macroscopic materials. Also the oxygen which has two lone pairs of electrons can form two hydrogen bonds with hydrogen atoms. Hydrogen bond strengths range from 4 kJ to50 kJ per mole of hydrogen bonds. It results fromthe attractive force between a hydrogen atom covalently bonded toa very electronegative atom such as a N O or F atom and another veryelectronegative atom.
 Source: slidetodoc.com
Source: slidetodoc.com
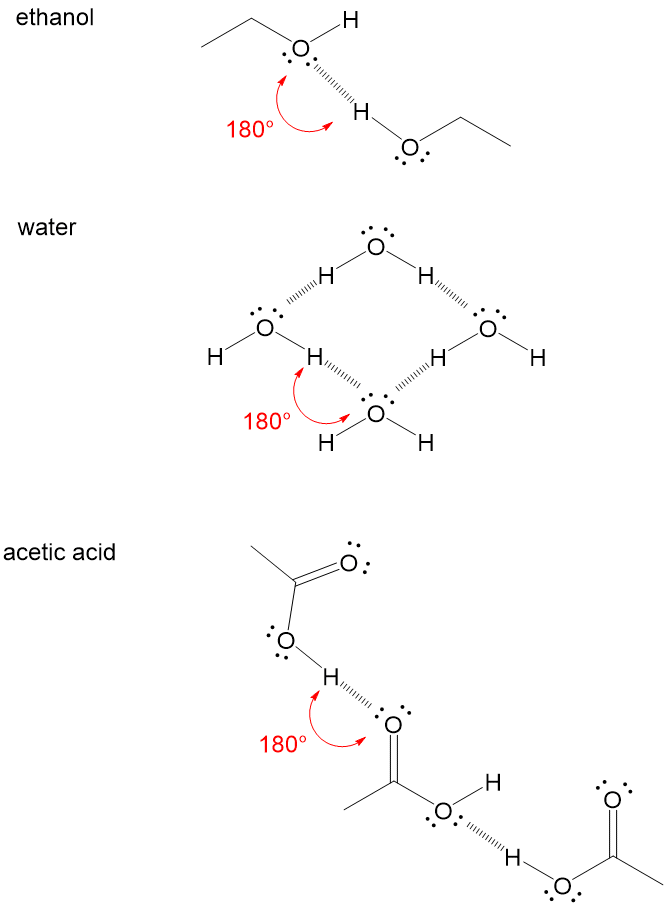
For example several H-F molecules are associated by hydrogen bond s as. The attractive force which binds hydrogen atom of one molecule with electronegative atom such as fluorine oxygen and nitrogen of another molecule generally of the same substance is known as hydrogen bond. In molecules containing N-H O-H or F-H bonds the large difference inelectronegativity between the H atom and the N O or F atom leads to ahighly polar. It results fromthe attractive force between a hydrogen atom covalently bonded toa very electronegative atom such as a N O or F atom and another veryelectronegative atom.
 Source: pinterest.com
Source: pinterest.com
Atom - atom - Atomic bonds. Hydrogen bond acceptor and the hydrogen itself can be as short as 18-19 Å well below the sum of the atomic radii eg. A hydrogen atom in a molecule forms a bond with any atom. The partially positive hydrogen atom of one molecule is then attracted to the oxygen atom of a nearby water molecule see Figure below.
 Source: studyorgo.com
Source: studyorgo.com
As these three atoms are the only ones with sufficient electronegativity that when bound to hydrogen can form hydrogen bonds. Each hydrogen atom can form a hydrogen bond with a nitrogen fluorine or oxygen atom. The bond between Mg and Cl in MgCl2 c. It results fromthe attractive force between a hydrogen atom covalently bonded toa very electronegative atom such as a N O or F atom and another veryelectronegative atom.
Each hydrogen atom can form a hydrogen bond with a nitrogen fluorine or oxygen atom.
The hydrogen atoms are bound to the highly electronegative oxygen atom which also possesses two lone pair sets of electrons making for a very polar bond. In molecules containing N-H O-H or F-H bonds the large difference inelectronegativity between the H atom and the N O or F atom leads to ahighly polar. As these three atoms are the only ones with sufficient electronegativity that when bound to hydrogen can form hydrogen bonds. You know that the Lennard-Jones potential rises with 1r12 if we get closer than the Van der Vaals radii. The bond between Na and Cl in salt e.
 Source: qsstudy.com
Source: qsstudy.com
Two oxygen atoms c. Hydrogen bond strengths range from 4 kJ to50 kJ per mole of hydrogen bonds. Also the oxygen which has two lone pairs of electrons can form two hydrogen bonds with hydrogen atoms. Each hydrogen atom can form a hydrogen bond with a nitrogen fluorine or oxygen atom. The bond between the H of one water molecule and the O of another water molecule b.
As these three atoms are the only ones with sufficient electronegativity that when bound to hydrogen can form hydrogen bonds.
Atom - atom - Atomic bonds. The nitrogen atom is called the hydrogen bond acceptor because it is accepting the hydrogen from the oxygen. The elements that usually participate in hydrogen bonds are nitrogen oxygen and fluorine. Which of the following is an example of a hydrogen bond.
 Source: qsstudy.com
Source: qsstudy.com
The bond between C. 12Å for hydrogen and 15Å for oxygen and nitrogen. The bond between Na and Cl in salt e. Water shown above is a molecule which can exhibit hydrogen bonding.
 Source: pinterest.com
Source: pinterest.com
Water shown above is a molecule which can exhibit hydrogen bonding. A hydrogen atom forms a covalent bond with another atom. A hydrogen atom bonded to F O or N is attracted to an electron pair. This sums to four hydrogen bonds per water molecule.

The bond between two hydrogen atoms d. Once the way atoms are put together is understood the question of how they interact with each other can be addressedin particular how they form bonds to create molecules and macroscopic materials. You know that the Lennard-Jones potential rises with 1r12 if we get closer than the Van der Vaals radii. Hydrogen bond is formed only by the three highly electronegative elements- fluorine oxygen and nitrogen.
For example several H-F molecules are associated by hydrogen bond s as.
Atom - atom - Atomic bonds. It results fromthe attractive force between a hydrogen atom covalently bonded toa very electronegative atom such as a N O or F atom and another veryelectronegative atom. In other words hydrogen bonding occ. The nitrogen atom is called the hydrogen bond acceptor because it is accepting the hydrogen from the oxygen. Atom - atom - Atomic bonds.
 Source: toppr.com
Source: toppr.com
The bond between the H of one water molecule and the O of another water molecule b. Also the oxygen which has two lone pairs of electrons can form two hydrogen bonds with hydrogen atoms. The bond between the H of one water molecule and the O of another water molecule b. Hydrogen bond is formed only by the three highly electronegative elements- fluorine oxygen and nitrogen. Hydrogen bond acceptor and the hydrogen itself can be as short as 18-19 Å well below the sum of the atomic radii eg.
A hydrogen bond is a primarily electrostatic force of attraction between a hydrogen atom which is covalently bound to a more electronegative atom or group and another electronegative atom bearing a lone pair of electronsthe hydrogen bond acceptor.
The elements that usually participate in hydrogen bonds are nitrogen oxygen and fluorine. Due to the large electronegativity difference strong dipoles are created on either end of the molecule as shown below. Two hydrogen atoms b. A hydrogen atom and an oxygen atom.
 Source: slidetodoc.com
Source: slidetodoc.com
A hydrogen atom and an oxygen atom. The elements that usually participate in hydrogen bonds are nitrogen oxygen and fluorine. These can then attract one another and create substances with higher boiling points. The partially positive hydrogen atom of one molecule is then attracted to the oxygen atom of a nearby water molecule see Figure below. In the picture of two water molecules at lower right the oxygen of the water molecule B is the hydrogen bond donor.
 Source: qsstudy.com
Source: qsstudy.com
The bond between Na and Cl in salt e. A hydrogen bond can form between_____ adjacent to each other. A hydrogen atom and an oxygen atom. In molecules containing N-H O-H or F-H bonds the large difference inelectronegativity between the H atom and the N O or F atom leads to ahighly polar. Hydrogen bond acceptor and the hydrogen itself can be as short as 18-19 Å well below the sum of the atomic radii eg.
 Source: pinterest.com
Source: pinterest.com
Hydrogen bond strengths range from 4 kJ to50 kJ per mole of hydrogen bonds. Two oxygen atoms c. So hydrogen bonding is possible only in those compounds in which the hydrogen atom is directly bonded to fluorine oxygen or nitrogen. The first way gives rise to what is called an ionic bond. In molecules containing N-H O-H or F-H bonds the large difference inelectronegativity between the H atom and the N O or F atom leads to ahighly polar.
This site is an open community for users to share their favorite wallpapers on the internet, all images or pictures in this website are for personal wallpaper use only, it is stricly prohibited to use this wallpaper for commercial purposes, if you are the author and find this image is shared without your permission, please kindly raise a DMCA report to Us.
If you find this site beneficial, please support us by sharing this posts to your favorite social media accounts like Facebook, Instagram and so on or you can also bookmark this blog page with the title which of the following atoms can attract a hydrogen atom in a hydrogen bond by using Ctrl + D for devices a laptop with a Windows operating system or Command + D for laptops with an Apple operating system. If you use a smartphone, you can also use the drawer menu of the browser you are using. Whether it’s a Windows, Mac, iOS or Android operating system, you will still be able to bookmark this website.





